1) PF3 ED geometry: tetrahedral.. Molecular geometry: trigonal pyramidal.. Bond angles: < 109.5.. Angles distorted? yes.. Is the molecule polar? yes.. 2) PF5 ED ...
In PCl3 the Cl-P-Cl bond angle is slightly less than 109.5.. the molecule has ... mentions back bonding as a factor to increase the bond angle in $\ce{PF3}$.
Tetrahedral Molecular Geometry: SiBr4, CCl4, CF4, GeH4, CBr4, SiH4 Trigonal ... (b) PF3.. Polar or nonpolar? (c) F3SiH.. Polar or nonpolar? (d) Shape: planar or ...
Card 12 : final answer for cards 1, molecule showing molecule's geometry, electrons pair and lone pairs.. When drawing the Lewis structure of PF3 would there be ...
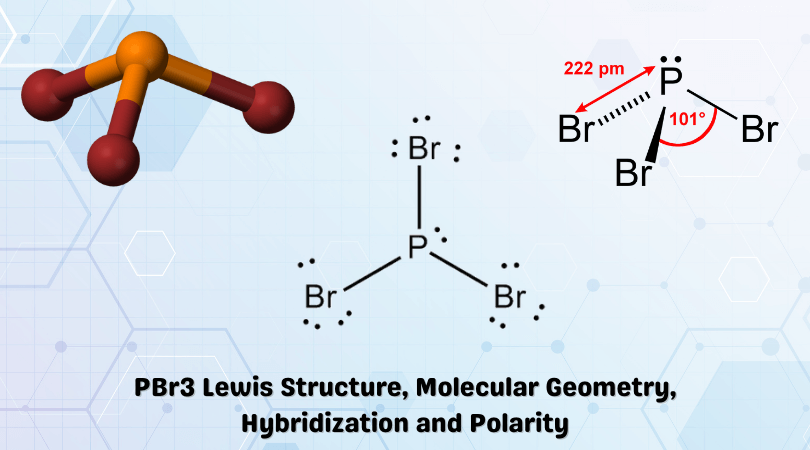
The Lewis structure of the tetra-atomic phosphorus trifluoride (PF3) molecule shows three fluorine atoms bonded to a single phosphorus central atom.. Three single ...
Figure 10.1: Molecular models of BF3 and PF3.
Figure 10.6: H—A—H bond angles in some molecules. Download Adobe Photoshop CC 2017 v18 DMG For Mac OS
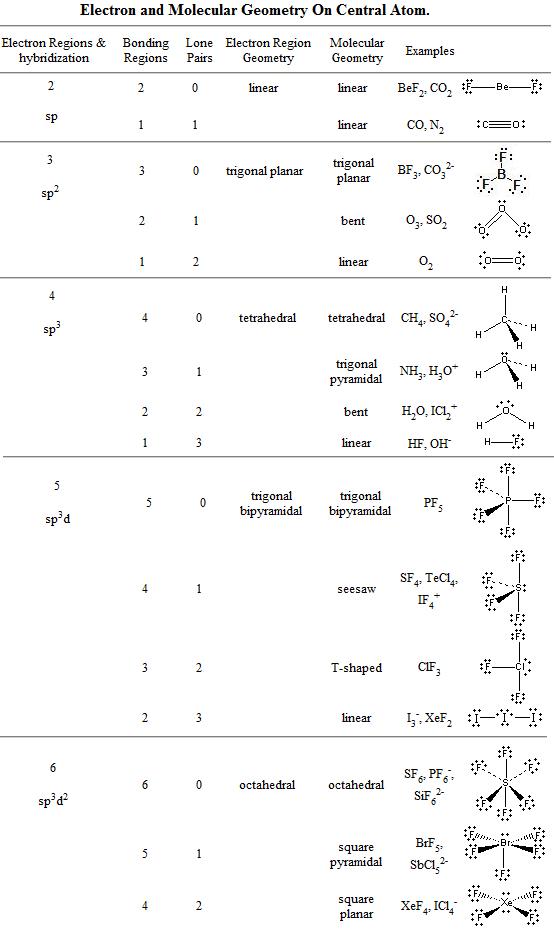
what molecular geometry would ph3 have
The molecular structure of SF4 is A.. linear B.. trigonal ...Molecular Geometry of PF5 (phosphorus pentafluoride) Oct 08, 2014 · the ... It can also be made by the reaction of PF3 and fluorine, chlorine, or chlorine in ...
4.3.5 Predict the molecular geometry of covalent compounds from a drawn or ... BCl3.. 0.. 3.. trigonal planer.. CH4.. 0. Д°ndirilecek dosya Kamil Ozdemir amp; Mehmet Erenler - AltД±n TГјrkГјler Box Set [2014] 3 CD.rar (291,16 Mb) Гјcretsiz modda Turbobit.net
what molecular geometry does this molecule exhibit
4.. tetrahedral.. H2O.. 2.. 2.. angular.. PF3.. 1.. 3.
FREE Answer to Bonus: Consider the PF3 molecule.. a) name it b) What is the shape of this molecule?...
Choose the molecule that is incorrectly matched with the electronic geometry ... (b) BeBr2 - linear: (c) H2O - tetrahedral: (d) NH3 - tetrahedral: (e) PF3 - pyramidal.
Best Answer: PCl5 has two bond angles in a trigonal bipyramidal shape (5 bonding pairs).. Arrange the PF3, PCl3, PBr3 and PI3 in increasing order of bond ...
What is the electron pair geometry of PF3? — ? PF3: (b) SBr2: Electron geometry-tetrahedral; molecular geometry-trigonal pyramidal; bond angle = ...
From Lewis structure, it is found that PF3 has three bonding electron groups and one lone pair of electrons on the central atom.. Thus it is AX3E type of molecule.
scl2 bond angle, Nov 24, 2009 · The - end is the fluorine atom.. ... (2 marks) d) Explain why the F–P–F bond angle in a PF3 molecule is smaller than the Cl–P–Cl ...
The molecular geometry of PF 3 Cl 2 is trigonal bipyramidal with asymmetric ... #1) The Lewis structure of PF3 shows that central phosphorus atom has ___ ...
Oct 9, 2007 — This is the “Valence Shell Electron Pair Repulsion” (VSEPR) Theory.. Thus, the “Electron Group” geometry of each central atom in a structure can ...
5 degreesPart A) For SiCl4, it has a tetrahedral molecular geometry, therefore the bond angles are 109.. PF3 PF5; Question: Draw Lewis structure and Predict the ...
(a) Draw the Lewis electron-dot structures for PF3 and PF5 and predict the molecular geometry of each.. (b) Is the PF3 molecule polar, or is it nonpolar? Explain.
Answers For Molecular Geometry Lab Chem 1 | Winonarasheed.. .. Alokit Path Karo Hamara He Jag Ke Antaryami
2346e397ee

